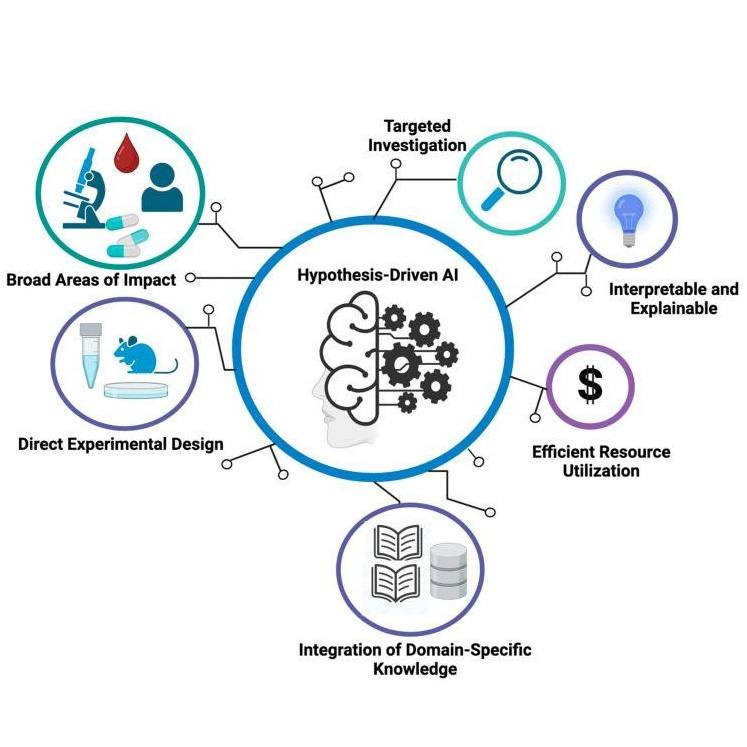
Medical Innovation

April 18, 2024
On March 15, in an auditorium packed with colleagues, students and supporters, 13 innovators gathered on the University of Minnesota campus for the 2024 Walleye[...]
April 4, 2024
April 2, 2024

February 28, 2024
February 26, 2024
Explore more topics
 Sign up
Sign up

Mayo Clinic Connect
An online patient support community