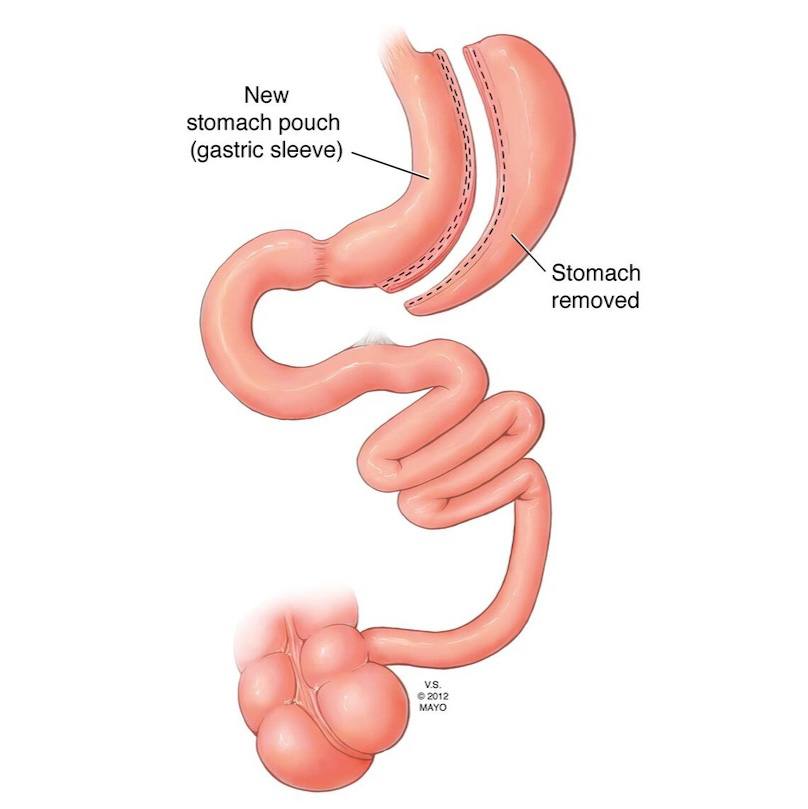
Latest news on Health Topics




Latest Medical Research

Discovery Science
April 30, 2024

Healthcare Delivery
April 29, 2024
Editor's Choice

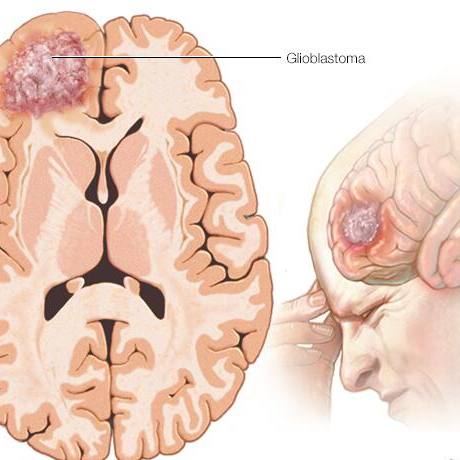

Explore Health Resource Topics
 Sign up
Sign up

Mayo Clinic Connect
An online patient support community